Endometriosis
is a puzzling disease, there are still many unanswered question surrounding its
origin, who gets it and why. In this way it makes studying endometriosis like
exploring the world in the 17th century, there is a sense of undiscovered
territory to explore, but rather than large wooden ships and subjugating
natives, we have theories and hypotheses to test with science. Some of those
theories prove worth exploring, some have a little, a lot, or no evidence to
support them. Creating new theories sometimes seems like muddying the water and
complicated an already complicated subject, but it is only by coming up with
and testing new ideas, we get closer to the truth (however, all ideas must be testable and all tests must be fair, objective and subject to
scrutiny by peer review, something pseudoscience always fails at).
With that in
mind I’m going to be doing a bit of theorising myself in this week’s blog post.
A big part of my research is examining the role that inflammation plays in
endometriosis, specifically what controls inflammation in endometriotic and
normal endometrial tissue. Part of this research involves looking into the
initial causes of inflammation, and one thing in particular has caught my
attention recently and that is bacteria.
Some types
of bacteria are essential for our health, without them we would become very ill
indeed and several of our organs have their own population of micro-organisms
that help them function correctly. The gut and the vagina are two good examples,
the gut in particular contains between 500 and 1000 different species of
bacteria, numbering in the trillions of individual micro-organisms. Interestingly,
the harmless bacteria that naturally populate the vagina actually help prevent
infections by more harmful bacterial types. But sometimes disruption of the
natural order of our internal microflora can lead to harmful effects.
Now I’m not
falling into the trap of confusing endometriosis with endometritis and I’m
certainly not suggesting that endometriosis is an infection or caused by an
infection. Rather bacteria may play a hitherto under-recognised role in
inflammation and the generation of painful symptoms in endometriosis.
How might
bacteria contribute to endometriosis? To clarify I’m talking about a specific
type of bacteria, called Gram-negative
bacteria, which is a group of bacteria including E.coli, one of the better
known species of bacteria. Why these organisms are important is because of
something they secrete, namely lipopolysaccharides (LPS). LPS are essentially
toxins the bacteria produce that are responsible for making you ill, but they are
also powerful stimulants for inflammation and this is the key point of interest.
To start
with we need to know which bacteria are normally present in the uterus and how
this differs in women with endometriosis. A study published in 2014
investigated this very phenomena and found that several bacteria types,
especially E.coli, were increased in the endometrium of women with
endometriosis (see the figure below)
The solid
white boxes represent the levels of micro-organisms found in the endometrium of
women without endo, the dashed boxes represent the levels in women with endo.
What we can
see from this is that there are several types of micro-organism found at increased
levels in the endometrium of women with endometriosis, in particular a large
increase in E.coli. Following on from this further studies found that
LPS was found at a much higher concentration in the menstrual fluid and peritoneal
fluid of women with endo as you can see if you click this link and look at figures C and D (the white
bars represent the levels of LPS in women without endo, the black bars are
women with endo).
The increase
in LPS in the menstrual fluid can be attributed to the increase in E.coli in
the endometrium, but what about the peritoneal fluid? This can be explained by
retrograde menstruation, where menstrual fluid is passed upwards through the
fallopian tubes and out into the peritoneal cavity. Retrograde menstruation is
a natural event in the female body during menstruation, occurring in as much as 90% of women
and it was long thought that this was the root cause of endo, with endometrial
cells shed during a period being passed into the peritoneal cavity where they
implant and grow. This theory has since fell from favour somewhat as more
evidence accumulates against it being the cause of endo, but there may be a
role for retrograde menstruation in endometriosis, just perhaps not what was
previously thought.
It seems plausible
then that increased levels of E.coli in the endometrium lead to higher
concentrations of LPS in the menstrual blood, which can get refluxed into the
peritoneal cavity by retrograde menstruation, increasing the concentration of
LPS in the peritoneal fluid, which can be in close/direct contact with
endometriotic lesions. The next step is to assess what can happen when
LPS interacts
with endometriosis.
Some studies have
looked into this and found that LPS can stimulate endometriotic cells to
release factors, like prostaglandin E2 (PGE2) and tumour necrosis factor (TNFα) which promote inflammation and stimulate pain
signals (PGE2 has been implicated
in a number of important facets for endometriosis survival including cell growth,
inhibition cell destruction and hormone production). This would lead to a heightened
inflammatory state around endometriotic lesions and in the normal endometrium, possibly
contributing to symptoms frequently associated with endometriosis, such as
chronic pelvic pain and excessively painful periods. The whole process can be summed
up on the diagram below.
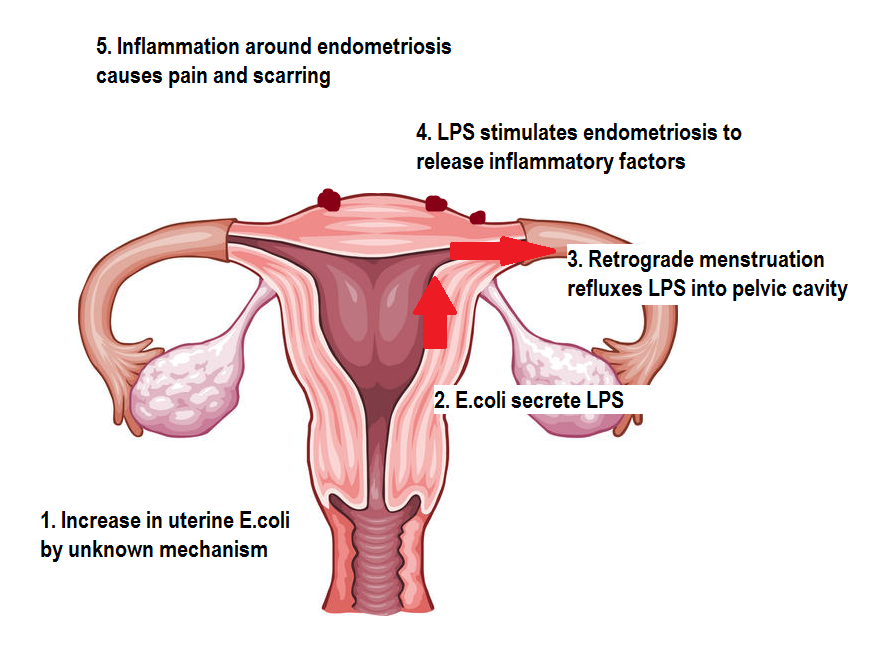 |
| Original image from https://edc2.healthtap.com/ht-staging/user_answer/reference_image/7081/large/Uterus.jpeg?1386669522 |
The final
question, which we don’t have an answer to yet, is why E.coli and other
micro-organisms are increased in the endometrium of women with endo. It may be due to alterations of vaginal
acidity leading to easier colonisation of the uterus by bacteria, or
dysfunction of the immune cells in the uterus of women with endo resulting in
them not being able to keep micro-organisms in check? Also, what effect would
restoring the normal balance of uterine micro-organisms have and how can this
be achieved? There are a lot of issues that need to be addressed here, but
hopefully this will be an area of investigation that yields positive results in
terms of relief from painful symptoms.
No comments:
Post a Comment
Note: only a member of this blog may post a comment.